![[Translate to Finnish:] [Translate to Finnish:]](/fileadmin/user_upload/Case_Studies/fixed_size_List_View/BioTechnique_interior.jpg)
[Translate to Finnish:]
BioPharm-yritys osoittaa todeksi MEGAcel® II -suodattimen luotettavuuden
- BioTechnique™ on yhdysvaltalainen erittäin voimakkaiden ja sytotoksisten, steriilien ruiskulla annettavien biolääkevalmisteiden valmistaja
- Sen tuotantolaitos sijaitsee Wisconsinin Madisonissa
- BioTechnique valmistaa:
- Nestemäiset ja kylmäkuivatut, steriilit, ruiskulla annettavat annosmuodot eläviä heikennettyjä viruksia, vasta-ainelääke-konjugaatteja, nanohiukkaisia ja sytotokseja
- Loppusterilointiin sopimattomat tuotteet, joista aiheutuu erityisiä työympäristö- ja turvallisuushaittoja, asettavat niitä käsittelevän henkilöstön kovalle rasitukselle

Ilmankäsittely oli erityisen haasteellista BioTechniquen tuotantotiloissa, koska kaikki BioTechniquen tuotteet on valmistettava aseptisessa ympäristössä. Jos HEPA-suodatin oli jotenkin vahingoittunut eikä pienentänyt vähintään 99,97 %:lla elinkyvyttömien hiukkasten määrää, steriiliksi vaaditussa tuotteessa saattoi muodostua haitallista mikrobikasvustoa. Riskianalyysi osoitti mahdollisesti kriittisen vaikutuksen potilaille, jos käytettiin muita kuin steriilejä tuotteita. Tätä ei voitu hyväksyä.
HEPA- suodatusluokan rike voidaan havaita tuotteen AQL-steriiliysnäytteen ottamisen, vaaditun ympäristövalvonnan, tuotenäytteen tai kahden vuoden välein tehtävän HEPA-sertifiointiprosessin aikana. Tarvittiin kuitenkin parempi tekninen ratkaisu, jotta voitiin varmistaa, että BioTechnique pystyi ylläpitämään korkeimpia mahdollisia standardeja. Kun tuote on valmistettu, täytetty, tarkastettu, merkitty, pakattu, testattu ja päästetty markkinoille, ei ole enää mahdollisuuksia havaita laatuongelmia. Arvioituaan mahdollisen potilasturvallisuusriskin BioTechniquen johto antoi teknisen suunnittelun osastolle valtuudet etsiä ratkaisu, jolla parannetaan valmistusprosessin luotettavuutta ja tuotteen laatua ja samalla pienennetään tuotantotilan energiankulutusta.
Yritys käynnisti loppuvuodesta 2014 Lean Six Sigma -projektin, jonka tarkoitus oli parantaa tuotantotilojen luotettavuutta ja pienentää energiakustannuksia, joita koitui luokan A (ISO 5), luokan B (ISO 6), luokan C (ISO 7) ja luokan D (ISO 8) alueiden ylläpidosta. Kansalliset ja paikalliset energiahankkeet, kuten State of California Title 24 -ohjelma, ovat kannustaneet useita lääkeyrityksiä kuten Biotechnique käynnistämään energiansäästöhankkeita.
BioTechnique käynnisti teknisen tutkimuksen, jolla pyrittiin määrittämään pääomatuotto, kun hankittaisiin uusi PAO-yhteensopiva ePTFE-suodatin, verrattuna perinteiseen lasikuitumateriaaliin. Tutkimuksen tarkoitus oli määrittää suodatinpakin optimaalinen syvyys asennettavalle ePTFE-suodatinmateriaalille, suhteessa vanhaan suodattimen mikrolasikuitumateriaaliin.
Jotta tuloksissa ei olisi tilastollista vinoumaa BioTechnique rakensi koepenkin, joka mahdollisti testauksen valvotuissa olosuhteissa ja varmisti, että testausmenetelmä oli tilastollisesti kelvollinen ja toistettavissa ja että tulokseksi saataisiin olennaisia tietoja, joiden avulla voitaisiin tehdä päteviä johtopäätöksiä. Tutkimuksessa tehtiin testejä, jotka suoritettiin kolmeen kertaan, joka kerta kolmea kumpaakin suodatintyyppiä käyttäen, mukaan lukien kaksi perinteistä HEPA-mikrolasikuitusuodatinta ja 50 mm ja 75 mm ePTFE HEPA -suodatinta. Tutkimuksessa käytettiin siis 36 eri testiä, millä saavutettiin tilastollisesti merkittävä tutkimus.
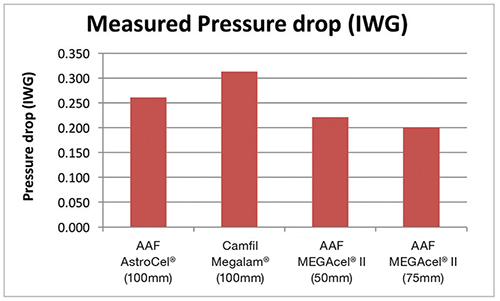
Tässä kaaviossa on esitetty helmikuussa 2015 neljästä suodatintyypistä kerätyt tiedot. 50 mm:n ePTFE-suodattimen painehäviö oli noin 30 % vähemmän kuin 100 mm:n HEPA-mikrolasikuitusuodattimella. 75 mm:n ePTFE-suodattimen painehäviö oli noin 36 % vähemmän kuin tavanomaisella 100 mm:n HEPA-mikrolasikuitusuodattimella.
BioTechnique laski ennustetun vuotuisen kokonaissäästön ePTFE HEPA -suodattimien pienemmän painehäviön ansiosta olevan 9,350 kWh.
Lokakuussa 2015 kaksi kilpailevaa suodatinsettiä (AAF MEGAcel® II ja Kilpailijan) asennettiin kahteen erilliseen ISO 5 -kriittiseen tilaan ja testattiin FDA-sertifiointivaatimusten mukaan useita kertoja.
Koko testikäytön ajan MEGAcel II -suodattimien vaste oli 100-prosenttinen ensimmäisen vaiheen onnistuminen, kun taas kilpailijan suodattimet osoittivat valitettavaa 10,8 %:n vikaantumisastetta, minkä johdosta suodattimet oli vaihdettava ja uusintatestattava.
Tulokset vahvistivat paitsi, että AAF:n MEGAcel II lääkeluokan ePTFE-suodatinteknologia on PAO-yhteensopivaa, sen suodatusteho on lisäksi korkeinta tasoa kriittisillä alueilla.
Koska MEGAcel II -suodattimet olivat merkittävästi kevyempiä, yksi henkilö pystyy tekemään asennuksen, kun HEPA-mikrolasikuitusuodattimen asennuksessa tarvitaan kahta henkilöä. Tämä työkustannussäästö on erityisen tärkeä silloin, kun asennetaan merkittäviä suodatinmääriä. Suodatinta voidaan pitää toisessa kädessä ja ruuvitalttaa toisessa, mikä mahdollistaa nopeamman asennuksen. Lisäetuna on asennuksen huolettomuus—vahvempi ja vankempi suodatin tarkoittaa, että suodattimen vahingoittumista ei tarvinnut varoa.

Testit osoittivat, että ePTFE-materiaalista valmistetut HEPA-suodattimet olivat merkittävästi vahvempia ja luotettavampia kuin perinteinen mikrolasikuitumateriaali.
- AAF:n MEGAcel II -suodattimet selvisivät kilpailussa ylivoimaisella 100-prosenttisesti vuotovapaalla sertifioinnilla.
- ePTFE-suodattimet vähensivät suodattimien vikaantumisia ja ympäristövalvontatehtäviä, jonka ansiosta tuotantoseisokit vähenivät ja työntekijäkustannukset pienenivät.
- ePTFE-suodattimien käytöllä BioTechniquen uudelleensertifiointien väli pidentyi ja puhdastilan käyttöaika kasvoi.
- MEGAcel II -suodattimien keveys mahdollisti nopeamman asennuksen—työvoimakustannuksiin huomattava pienennys.
- ePTFE-materiaalin painehäviö on merkittävästi pienempi kuin perinteisen mikrolasikuitumateriaalin, minkä ansiosta saavutetaan merkittäviä energiansäästöjä luokan A ja luokan B puhdastiloissa.
- BioTechnique™ on yhdysvaltalainen erittäin voimakkaiden ja sytotoksisten, steriilien ruiskulla annettavien biolääkevalmisteiden valmistaja
- Sen tuotantolaitos sijaitsee Wisconsinin Madisonissa
- BioTechnique valmistaa:
- Nestemäiset ja kylmäkuivatut, steriilit, ruiskulla annettavat annosmuodot eläviä heikennettyjä viruksia, vasta-ainelääke-konjugaatteja, nanohiukkaisia ja sytotokseja
- Loppusterilointiin sopimattomat tuotteet, joista aiheutuu erityisiä työympäristö- ja turvallisuushaittoja, asettavat niitä käsittelevän henkilöstön kovalle rasitukselle

Ilmankäsittely oli erityisen haasteellista BioTechniquen tuotantotiloissa, koska kaikki BioTechniquen tuotteet on valmistettava aseptisessa ympäristössä. Jos HEPA-suodatin oli jotenkin vahingoittunut eikä pienentänyt vähintään 99,97 %:lla elinkyvyttömien hiukkasten määrää, steriiliksi vaaditussa tuotteessa saattoi muodostua haitallista mikrobikasvustoa. Riskianalyysi osoitti mahdollisesti kriittisen vaikutuksen potilaille, jos käytettiin muita kuin steriilejä tuotteita. Tätä ei voitu hyväksyä.
HEPA- suodatusluokan rike voidaan havaita tuotteen AQL-steriiliysnäytteen ottamisen, vaaditun ympäristövalvonnan, tuotenäytteen tai kahden vuoden välein tehtävän HEPA-sertifiointiprosessin aikana. Tarvittiin kuitenkin parempi tekninen ratkaisu, jotta voitiin varmistaa, että BioTechnique pystyi ylläpitämään korkeimpia mahdollisia standardeja. Kun tuote on valmistettu, täytetty, tarkastettu, merkitty, pakattu, testattu ja päästetty markkinoille, ei ole enää mahdollisuuksia havaita laatuongelmia. Arvioituaan mahdollisen potilasturvallisuusriskin BioTechniquen johto antoi teknisen suunnittelun osastolle valtuudet etsiä ratkaisu, jolla parannetaan valmistusprosessin luotettavuutta ja tuotteen laatua ja samalla pienennetään tuotantotilan energiankulutusta.
Yritys käynnisti loppuvuodesta 2014 Lean Six Sigma -projektin, jonka tarkoitus oli parantaa tuotantotilojen luotettavuutta ja pienentää energiakustannuksia, joita koitui luokan A (ISO 5), luokan B (ISO 6), luokan C (ISO 7) ja luokan D (ISO 8) alueiden ylläpidosta. Kansalliset ja paikalliset energiahankkeet, kuten State of California Title 24 -ohjelma, ovat kannustaneet useita lääkeyrityksiä kuten Biotechnique käynnistämään energiansäästöhankkeita.
BioTechnique käynnisti teknisen tutkimuksen, jolla pyrittiin määrittämään pääomatuotto, kun hankittaisiin uusi PAO-yhteensopiva ePTFE-suodatin, verrattuna perinteiseen lasikuitumateriaaliin. Tutkimuksen tarkoitus oli määrittää suodatinpakin optimaalinen syvyys asennettavalle ePTFE-suodatinmateriaalille, suhteessa vanhaan suodattimen mikrolasikuitumateriaaliin.
Jotta tuloksissa ei olisi tilastollista vinoumaa BioTechnique rakensi koepenkin, joka mahdollisti testauksen valvotuissa olosuhteissa ja varmisti, että testausmenetelmä oli tilastollisesti kelvollinen ja toistettavissa ja että tulokseksi saataisiin olennaisia tietoja, joiden avulla voitaisiin tehdä päteviä johtopäätöksiä. Tutkimuksessa tehtiin testejä, jotka suoritettiin kolmeen kertaan, joka kerta kolmea kumpaakin suodatintyyppiä käyttäen, mukaan lukien kaksi perinteistä HEPA-mikrolasikuitusuodatinta ja 50 mm ja 75 mm ePTFE HEPA -suodatinta. Tutkimuksessa käytettiin siis 36 eri testiä, millä saavutettiin tilastollisesti merkittävä tutkimus.

Tässä kaaviossa on esitetty helmikuussa 2015 neljästä suodatintyypistä kerätyt tiedot. 50 mm:n ePTFE-suodattimen painehäviö oli noin 30 % vähemmän kuin 100 mm:n HEPA-mikrolasikuitusuodattimella. 75 mm:n ePTFE-suodattimen painehäviö oli noin 36 % vähemmän kuin tavanomaisella 100 mm:n HEPA-mikrolasikuitusuodattimella.
BioTechnique laski ennustetun vuotuisen kokonaissäästön ePTFE HEPA -suodattimien pienemmän painehäviön ansiosta olevan 9,350 kWh.
Lokakuussa 2015 kaksi kilpailevaa suodatinsettiä (AAF MEGAcel® II ja Kilpailijan) asennettiin kahteen erilliseen ISO 5 -kriittiseen tilaan ja testattiin FDA-sertifiointivaatimusten mukaan useita kertoja.
Koko testikäytön ajan MEGAcel II -suodattimien vaste oli 100-prosenttinen ensimmäisen vaiheen onnistuminen, kun taas kilpailijan suodattimet osoittivat valitettavaa 10,8 %:n vikaantumisastetta, minkä johdosta suodattimet oli vaihdettava ja uusintatestattava.
Tulokset vahvistivat paitsi, että AAF:n MEGAcel II lääkeluokan ePTFE-suodatinteknologia on PAO-yhteensopivaa, sen suodatusteho on lisäksi korkeinta tasoa kriittisillä alueilla.
Koska MEGAcel II -suodattimet olivat merkittävästi kevyempiä, yksi henkilö pystyy tekemään asennuksen, kun HEPA-mikrolasikuitusuodattimen asennuksessa tarvitaan kahta henkilöä. Tämä työkustannussäästö on erityisen tärkeä silloin, kun asennetaan merkittäviä suodatinmääriä. Suodatinta voidaan pitää toisessa kädessä ja ruuvitalttaa toisessa, mikä mahdollistaa nopeamman asennuksen. Lisäetuna on asennuksen huolettomuus—vahvempi ja vankempi suodatin tarkoittaa, että suodattimen vahingoittumista ei tarvinnut varoa.

Testit osoittivat, että ePTFE-materiaalista valmistetut HEPA-suodattimet olivat merkittävästi vahvempia ja luotettavampia kuin perinteinen mikrolasikuitumateriaali.
- AAF:n MEGAcel II -suodattimet selvisivät kilpailussa ylivoimaisella 100-prosenttisesti vuotovapaalla sertifioinnilla.
- ePTFE-suodattimet vähensivät suodattimien vikaantumisia ja ympäristövalvontatehtäviä, jonka ansiosta tuotantoseisokit vähenivät ja työntekijäkustannukset pienenivät.
- ePTFE-suodattimien käytöllä BioTechniquen uudelleensertifiointien väli pidentyi ja puhdastilan käyttöaika kasvoi.
- MEGAcel II -suodattimien keveys mahdollisti nopeamman asennuksen—työvoimakustannuksiin huomattava pienennys.
- ePTFE-materiaalin painehäviö on merkittävästi pienempi kuin perinteisen mikrolasikuitumateriaalin, minkä ansiosta saavutetaan merkittäviä energiansäästöjä luokan A ja luokan B puhdastiloissa.

 FHK Polska
FHK Polska PureMedion Kft.
PureMedion Kft. Elfa spol. s r. o.
Elfa spol. s r. o. Ecotip d.o.o.
Ecotip d.o.o.![[Translate to Finnish:] [Translate to Finnish:]](/fileadmin/_processed_/c/f/csm_hatting_agriculture_1b6a249990.jpg)
![[Translate to Finnish:] [Translate to Finnish:]](/fileadmin/_processed_/c/0/csm_microelectronics_smaller_dec03e69e3.jpg)
